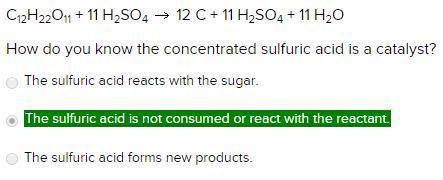
C12H22O11 + 11 H2SO4 12 C + 11 H2SO4 + 11 H2O
How do you know the concentrated sulfuric...

Chemistry, 25.03.2020 05:01 HNesmith16
C12H22O11 + 11 H2SO4 12 C + 11 H2SO4 + 11 H2O
How do you know the concentrated sulfuric acid is a catalyst?
The sulfuric acid reacts with the sugar.
The sulfuric acid is not consumed or react with the reactant.
The sulfuric acid forms new products.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 19:40
What type of electromagnetic waves does the human eye see as the colors red blue or green a visible light waves b radio waves c infrared waves d microwaves
Answers: 1

Chemistry, 22.06.2019 21:50
Given the data below for the reaction, 2 a + 2 b + 4 c => d + e + 3 f, the reaction is fill in the [ ] order in a, fill in the [ ] order in b, fill in the [ ] order in c and fill in the [ ] order overall. (use the words "first, second, third, fourth" to fill each blank)experimentinitial conc of a, mol/l initial conc of b, mol/l initial conc of c, mol/l initial rate, mol/l.s1 0.1 0.1 0.2 2 x 10-32 0.2 0.3 0.2 6 x 10-33 0.3 0.1 0.2 2 x 10-34 0.4 0.3 0.4 1.2 x 10-2
Answers: 2

Chemistry, 23.06.2019 10:10
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10-3 m and k for the dissociation is 1.86x10-5. ch3cooh(aq)+h2o(l)+> h3o+(aq)+ch3coo-(aq) show me how to get the answer.
Answers: 3
You know the right answer?
Questions



History, 27.05.2020 12:57


Law, 27.05.2020 12:57




English, 27.05.2020 12:57




Mathematics, 27.05.2020 12:57

Mathematics, 27.05.2020 12:57






Social Studies, 27.05.2020 12:57