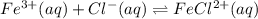Chemistry, 24.03.2020 23:08 tstaples02
Use Le Chatelier’s principle to predict how the equilibrium concentration of the FeCl2+ ion will change when an aqueous solution of silver (I) nitrate, AgNO3, is added to the exothermic reaction below. Fe3+(aq) + Cl-(aq) → FeCl2+(aq)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:00
What does earth’s rotation on its axis cause? the tides night and day passing of years phases of the moon
Answers: 1

Chemistry, 22.06.2019 03:30
In this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced?
Answers: 1

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1

Chemistry, 22.06.2019 18:20
Which reason best explains why metals are malleable? a)because they have delocalized electrons b)because they have localized electrons c)because they have ionic bonds d)because they have rigid bonds
Answers: 2
You know the right answer?
Use Le Chatelier’s principle to predict how the equilibrium concentration of the FeCl2+ ion will cha...
Questions

Mathematics, 09.10.2019 19:20



Mathematics, 09.10.2019 19:20

Mathematics, 09.10.2019 19:20

History, 09.10.2019 19:20














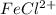 will decrease.
will decrease.