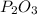The answers for the following sums is given below.
1.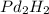
2.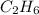
3.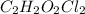
4.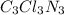
5.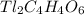
6.
7. 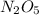
8.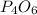
9.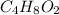
Explanation:
1.Given:
Molar mass=216.8g
Molecular formula=Pd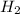
we know;
Molecular formula=n(Empirical formula)
molecular weight of palladium(Pd)=106.4u
molecular weight of hydrogen(H)=1u
Molar mass of Pd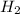 :
:
Pd=106.4×1=106.4u
H=1×2=2
molar mass of Pd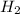 =106.4+2=108.4
=106.4+2=108.4
n=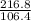
n=2
Molecular formula=2(Pd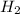 )
)
Molecular formula=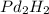
Therefore the molecular formula of the compound is 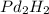
2. Given:
Molar mass=30.0g
Molecular formula=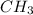
we know;
Molecular formula = n (Empirical formula)
molecular weight of Carbon(C)=12.01u
molecular weight of hydrogen(H)=1u
Molar mass of 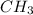 :
:
C=12.01 × 1 = 12.01u
H=1 × 3 = 3u
molar mass of 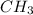 =12.01 + 3 =15.01u
=12.01 + 3 =15.01u
n=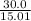
n=2
Molecular formula=2(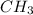 )
)
Molecular formula=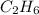
Therefore the molecular formula of the compound is 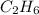
3. Given:
Molar mass=129g
Molecular formula=CHOCl
we know;
Molecular formula = n (Empirical formula)
molecular weight of Carbon(C)=12.01u
molecular weight of hydrogen(H)=1u
molecular weight of oxygen(O)=16.00u
molecular weight of chlorie(Cl)=35.5u
Molar mass of CHOCl:
C=12.01 × 1 = 12.01u
H=1 × 1 = 1u
O=16.00×1=16.00u
Cl=35.5×1=35.5u
molar mass of CHOCl=12.01+1+16.00+35.5=64.5u
n=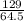
n=2
Molecular formula=2(CHOCl)
Molecular formula=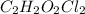
Therefore the molecular formula of the compound is 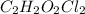
5. Given:
Molar mass=577g
Molecular formula=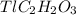
we know;
Molecular formula = n (Empirical formula)
molecular weight of Carbon(C)=12.01u
molecular weight of Thallium(Tl)=204.3u
molecular weight of hydrogen(H)=1u
molecular weight of oxygen(O)=16.00u
Molar mass of 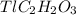 :
:
C=12.01 × 2= 24.02u
Tl=204.3×1=204.3u
H=1×2=2u
O=16.00×3=48.00
molar mass of 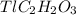 =204.3+24.02+1+48.00=278.32u
=204.3+24.02+1+48.00=278.32u
n=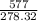
n=2
Molecular formula=2 (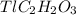 )
)
Molecular formula=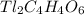
Therefore the molecular formula of the compound is 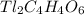
4. Molar mass=184.5g
Molecular formula=CClN
we know;
Molecular formula = n (Empirical formula)
molecular weight of Carbon(C)=12.01u
molecular weight of Nitrogen(N)=14u
molecular weight of chlorine(Cl)=35.5u
Molar mass of CClN:
C=12.01 × 1 = 12.01u
N=1×14=14U
Cl=35.5×1=35.5u
molar mass of CClN=12.01+14+35.5=61.5u
n=
n=3
Molecular formula=3 (CClN)
Molecular formula=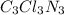
Therefore the molecular formula of the compound is 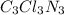
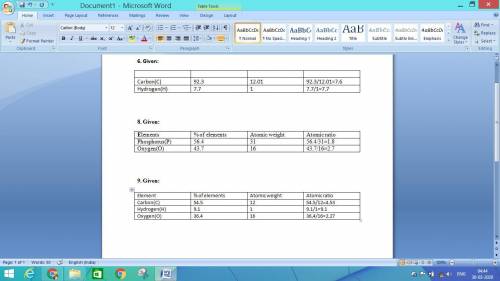
6. For the table refer the attached file.
Simplest ratio of elements:
Carbon=8
Hydrogen=8
Empirical formula=
Molecular formula =
Molar mass of  :
:
molecular weight of carbon=12.04u
molecular weight of hydrogen=1u
C=8×12.01=96.08u
H=1×8=8u
molar mass of  =96.08+8=104.08u
=96.08+8=104.08u
n=104.08÷78.0
n=1
Molecular formula = n(Empirical formula)
Molecular formula = 1( )
)
Molecular formula =
Therefore the molecular formula of a compound is 
7. Given:
mass of oxide of nitrogen=108g
mass of nitrogen=4.02g
mass of oxygen=11.48g
moles of nitrogen=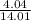 = 0.289 moles
= 0.289 moles
moles of oxygen=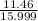 =0.716 moles
=0.716 moles
We divide through by the lowest molar quantity to give an empirical formula of 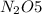 .
.
Now the molecular formula is multiple of the empirical formula.
So,
108 = n × (2×14.01 + 5×15.999)
Clearly,n=1, and the molecular formula is 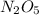 .
.
8.For the table refer the attached file.
Simplest ratio of elements:
Phosphorus=2
Oxygen=3
We know;
Empirical formula=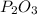
molecular formula= 2(Empirical formula)
Molecular formula =2(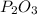 )
)
Molecular formula =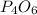
Therefore the molecular formula of the compound is 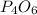
9. For the table refer the attached file.
Simplest ratio of elements:
Carbon=2
Hydrogen=9
Oxygen=2
We know;
Empirical formula =
Molecular formula = 2(Empirical formula)
Molecular formula =2( )
)
Molecular formula =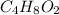
Therefore the molecular formula of the compound is 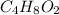



























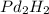
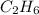
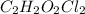
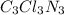
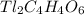

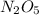
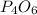
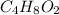
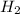
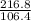
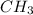
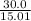
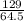
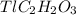
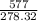

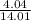 = 0.289 moles
= 0.289 moles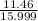 =0.716 moles
=0.716 moles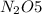 .
.