
A solution is prepared by adding 100 mL of 1.0 M HC₂H₃O₂(aq) to 100 mL of 1.0 M NaC₂H₃O₂(aq). The solution is stirred and its pH is measured to be 4.73. After 3 drops of 1.0 M HCl are added to the solution, the pH of the solution is measured and is still 4.73. Which of the following equations represents the chemical reaction that accounts for the fact that acid was added but there was no detectable change in pH?
(A)
(B)
(C)
(D)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Atrain travels 74 kilometers in 3 hours, and then 81 kilometers in 5 hours. what is its average speed?
Answers: 2

Chemistry, 22.06.2019 09:00
What is the percentage composition of carbon in the compound ch4
Answers: 1

Chemistry, 22.06.2019 09:40
Which diagram shows the correct way to represent an ionic compound of magnesium oxide?
Answers: 3

Chemistry, 22.06.2019 21:00
What is the chemical formula for the compound formed between sodium and flour one
Answers: 1
You know the right answer?
A solution is prepared by adding 100 mL of 1.0 M HC₂H₃O₂(aq) to 100 mL of 1.0 M NaC₂H₃O₂(aq). The so...
Questions


Mathematics, 09.03.2021 02:30


English, 09.03.2021 02:30

Mathematics, 09.03.2021 02:30

Mathematics, 09.03.2021 02:30



Social Studies, 09.03.2021 02:30

Mathematics, 09.03.2021 02:30

Mathematics, 09.03.2021 02:30

Health, 09.03.2021 02:30



Mathematics, 09.03.2021 02:30

Chemistry, 09.03.2021 02:30


Social Studies, 09.03.2021 02:30

Mathematics, 09.03.2021 02:30

Mathematics, 09.03.2021 02:30

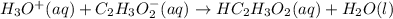 represents the chemical reaction that accounts for the fact that acid was added but there was no detectable change in pH.
represents the chemical reaction that accounts for the fact that acid was added but there was no detectable change in pH.

