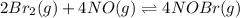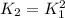Chemistry, 24.03.2020 17:51 rustalex6045
Consider the equilibrium reaction and its equilibrium constant expression. Br 2 ( g ) + 2 NO ( g ) − ⇀ ↽ − 2 NOBr ( g ) K = [ NOBr ] 2 [ Br 2 ] [ NO ] 2 For the reaction 2 Br 2 ( g ) + 4 NO ( g ) − ⇀ ↽ − 4 NOBr ( g ) select the equilibrium constant expression.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:30
Nebulae are enormous clouds in outer space. they are made mostly of hydrogen gas, helium gas, and dust. some nebulae glow brightly, while others do not. the stars that people see are huge, bright balls of glowing gas. they are made mostly of hydrogen and helium. which statement correctly describes other ways in which nebulae and stars are different? a. stars can form inside a nebula but a nebula can never be produced by any star. b. a star always has a higher density than a nebula. c. stars can never form inside a nebula but a nebula can be produced by any star. d. a nebula always has a higher density than a star.
Answers: 3

Chemistry, 22.06.2019 23:00
Which of your 24 wells had indications that a chemical reaction occurred? how were you able to tell that a chemical reaction occurred? which of your 24 wells had indications that a physical reaction occurred? how were you able to tell that a physical reaction occurred? report on both mixing and evaporation. make a general statement about whether your hypotheses were validated or rejected. must your hypotheses be correct for this to be a successful laboratory?
Answers: 3

Chemistry, 23.06.2019 00:30
Balance the following reaction. as2s3 + 9o2 → 2as2o3 + so2
Answers: 2

Chemistry, 23.06.2019 01:30
Concentrations expressed as a percent by mass are useful when the solute is a a. liquid b. gas c. solid
Answers: 1
You know the right answer?
Consider the equilibrium reaction and its equilibrium constant expression. Br 2 ( g ) + 2 NO ( g ) −...
Questions


Mathematics, 23.10.2020 23:30



History, 23.10.2020 23:30


Mathematics, 23.10.2020 23:30

Mathematics, 23.10.2020 23:30

Mathematics, 23.10.2020 23:30


Mathematics, 23.10.2020 23:30


Mathematics, 23.10.2020 23:30


English, 23.10.2020 23:30

Mathematics, 23.10.2020 23:30


Chemistry, 23.10.2020 23:30

History, 23.10.2020 23:30

![K_2=\frac{[NOBr]^4_{eq}}{[NO]^4_{eq}[Br]^2_{eq}}](/tpl/images/0560/9944/3b128.png)