
Consider the reaction 2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g) When 2 moles of Na react with water at 25°C and 1 atm, the volume of H2 formed is 24.5 L. Calculate the magnitude of work done in joules when 0.55 g of Na reacts with water under the same conditions. (The conversion factor is 1 L · atm = 101.3 J.)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3

Chemistry, 22.06.2019 00:30
What must happen before a body cell can begin mitotic cell division
Answers: 2

Chemistry, 23.06.2019 01:20
How can parts of a solution be separated by chromatography?
Answers: 1

Chemistry, 23.06.2019 08:30
Of element x has 22 protons, how many electrons does it have
Answers: 1
You know the right answer?
Consider the reaction 2Na(s) + 2H2O(l) → 2NaOH(aq) + H2(g) When 2 moles of Na react with water at 25...
Questions

Mathematics, 14.07.2019 06:00



Mathematics, 14.07.2019 06:00




Biology, 14.07.2019 06:00



History, 14.07.2019 06:00

Biology, 14.07.2019 06:00

Social Studies, 14.07.2019 06:00

Biology, 14.07.2019 06:00



Biology, 14.07.2019 06:00


Mathematics, 14.07.2019 06:00

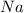
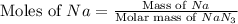
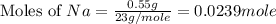
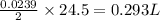 volume of hydrogen gas
volume of hydrogen gas

