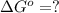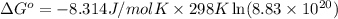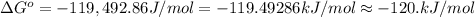In the activity, click on the E∘cell and Keq quantities to observe how they are related. Use this relation to calculate Keq for the following redox reaction that occurs in an electrochemical cell having two electrodes: a cathode and an anode. The two half-reactions that occur in the cell are
Cu2+(aq)+2e−→Cu(s) and Co(s)→Co2+(aq)+2e−
The net reaction is
Cu2+(aq)+Co(s)→Cu(s)+Co2+(aq)
Use the given standard reduction potentials in your calculation as appropriate. ( Keq=5.88*10^20)
In the activity, click on the Keq and ΔG∘ quantities to observe how they are related.
Calculate ΔG∘ using this relationship and the equilibrium constant (Keq) obtained in Part A at T=298K: Keq=5.88*10^20

Answers: 2


Another question on Chemistry


Chemistry, 23.06.2019 01:00
Which of the following is a physical change? a.burning a piece of wood b.sawing a piece of wood in half c.rust forming on an iron fence d.a copper roof changing color from orange to green
Answers: 1

Chemistry, 23.06.2019 01:30
Some molecular compounds, such as hcl, ionize completely in solution. for molecular compounds such as h2co3, most molecules do not ionize in solution. which describes the properties of these two solutes? a. hcl and h2co3 have the same effect on the properties of the solution. b. hcl raises the freezing point of water more than h2co3 does. c. hcl raises the boiling point of water more than h2co3 does.
Answers: 2

Chemistry, 23.06.2019 02:00
Scientists are often interested in knowing the molar heat of combustion – the heat released during the combustion of one mole of a substance. use the periodic table to find molar masses. how many moles of ethanol are present in the sample?
Answers: 2
You know the right answer?
In the activity, click on the E∘cell and Keq quantities to observe how they are related. Use this re...
Questions




Arts, 09.04.2021 17:10




Mathematics, 09.04.2021 17:10


Mathematics, 09.04.2021 17:10





Mathematics, 09.04.2021 17:10

Mathematics, 09.04.2021 17:10



Biology, 09.04.2021 17:10

 .
.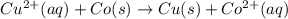
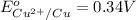
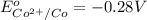
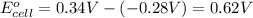
![E^o_{cell}=\frac{0.592}{n}\log[K_c]](/tpl/images/0559/8894/48eca.png)
![0.62 V=\frac{0.0592}{2}\log[K_c]](/tpl/images/0559/8894/f0d8e.png)
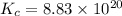
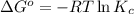
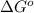 = standard Gibbs free energy =
= standard Gibbs free energy = 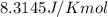
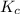 = Equilibrium constant
= Equilibrium constant