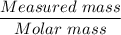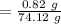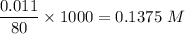Chemistry, 21.03.2020 08:05 lifeofabe214
A solution is composed of 1.15 mL of diethyl ether (C4H10O, density=0.7134 g/mL) dissolved in 78.85 mL of dichloromethane (CH2Cl2, density=1.3266 g/mL). What is the molarity of the solution?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:40
What kind of ion is contained in salts that produce an acidic solution? a positive ion that attracts a proton from water a positive ion that releases a proton to water a negative ion that attracts a proton from water a negative ion that releases a proton to water
Answers: 1

Chemistry, 21.06.2019 22:00
Which solution is an example of a nonelectrolyte? a. iodine in hexane b. sodium nitrate in waterc. acetic acid in waterd. hydrogen chloride in water
Answers: 2


Chemistry, 22.06.2019 11:00
Imagine that twenty i.u.’s of enzyme z were catalyzing the above reaction for one minute, under vmaxconditions, in a 3.00 ml assay volume. the assay is buffered with 20 mm phosphate buffer, ph 7.60. what will the ph be at the end of that one minute?
Answers: 2
You know the right answer?
A solution is composed of 1.15 mL of diethyl ether (C4H10O, density=0.7134 g/mL) dissolved in 78.85...
Questions


Geography, 21.08.2019 03:30




Social Studies, 21.08.2019 03:30

Social Studies, 21.08.2019 03:30

Mathematics, 21.08.2019 03:30

Mathematics, 21.08.2019 03:30




Biology, 21.08.2019 03:30




Chemistry, 21.08.2019 03:30

Social Studies, 21.08.2019 03:30


Mathematics, 21.08.2019 03:30