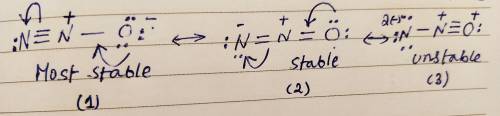Chemistry, 21.03.2020 03:16 winterblanco
Draw three resonance structures for N2O. This species has its three atoms bonded sequentially in the following fashion: N-O. Draw your resonance structures so that the atoms in them are bonded together in this order. Select the most important resonance structure for this species based on the formal charges on the atoms of the three resonance structures you have drawn. Now select the statement from the multiple choices which is true about this most important resonance structure. In the most important resonance structure of N2O :

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1

Chemistry, 22.06.2019 20:20
Which formula equation represents the burning of sulfur to produce sulfur dioxide? s(s) + o2(g) 4502(9) 2h2s(s) + 302(g) —> 2h20(0) + 2502(9) 4fes2+1102 —> 2fe2o3 + 8502 2802(g) + o2(9) v205 , 2503(9)
Answers: 1

Chemistry, 22.06.2019 23:00
What prefix multiplier is appropriate for reporting a measurement of 5.57 ×10−5 m?
Answers: 1

You know the right answer?
Draw three resonance structures for N2O. This species has its three atoms bonded sequentially in the...
Questions

English, 28.02.2021 20:20

History, 28.02.2021 20:20

History, 28.02.2021 20:20

Biology, 28.02.2021 20:20



Computers and Technology, 28.02.2021 20:20

Biology, 28.02.2021 20:20


Mathematics, 28.02.2021 20:20


Mathematics, 28.02.2021 20:20



Physics, 28.02.2021 20:20


Mathematics, 28.02.2021 20:20