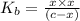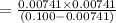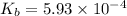Chemistry, 21.03.2020 03:01 bettybales1986
Aspirin (acetylsalicylic acid, C9H8O4) is a weak monoprotic acid. To determine its acid-dissociation constant, a student dissolved 2.00 g of aspirin in 0.600 L of water and measured the pH.
What was the Ka value calculated by the student if thepH of the solution was 2.62?
A 0.100 M solution of ethylamine (C2H5NH2) has a pH of 11.87.
Calculate the Kb for ethylamine.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
If you chip a tooth, most likely you will go to the dentist to have the missing material filled in. currently the material used to fill in teeth is a polymer that is flexible when put in, yet is hardened to the strength of a tooth after irradiation with blue light at a wavelength of 461 nm. what is the energy in joules for a photon of light at this wavelength?
Answers: 1

Chemistry, 22.06.2019 19:50
A2.5% (by mass) solution concentration signifies that there is a 2.5 % (by mass) solution concentration signifies that there is blank of solute in every 100 g of solution. of solute in every 100 g of solution
Answers: 3

Chemistry, 23.06.2019 02:30
Which statement best describes the liquid state of matter? a. it has definite shape but indefinite volume. b. it has definite shape and definite volume. c. it has indefinite shape and indefinite volume. d. it has indefinite shape but definite volume.
Answers: 1

You know the right answer?
Aspirin (acetylsalicylic acid, C9H8O4) is a weak monoprotic acid. To determine its acid-dissociation...
Questions







Biology, 29.10.2020 17:10






History, 29.10.2020 17:10







Mathematics, 29.10.2020 17:10

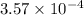 was the
was the 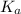 value calculated by the student.
value calculated by the student.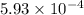 was the
was the 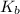 of ethylamine value calculated by the student.
of ethylamine value calculated by the student.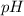 value of Aspirin solution = 2.62
value of Aspirin solution = 2.62![pH=-\log[H^+]](/tpl/images/0557/1305/cf945.png)
![[H^+]=10^{-2.62}=0.00240 M](/tpl/images/0557/1305/aef02.png)
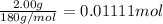
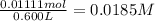
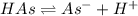
![K_a=\frac{[As^-][H^+]}{[HAs]}](/tpl/images/0557/1305/c8f9f.png) :
: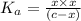
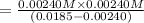
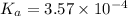
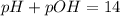
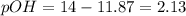
![pOH=-\log[OH^-]](/tpl/images/0557/1305/fe336.png)
![[OH^-]=10^{-2.13}=0.00741 M](/tpl/images/0557/1305/fa327.png)
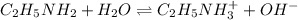
![K_b=\frac{[C_2H_5NH_3^{+}][OH^-]}{[C_2H_5NH_2]}](/tpl/images/0557/1305/63c9d.png) :
: