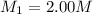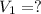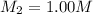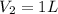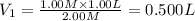A student wants to prepare 1.00 L of a 1.00 M solution of NaOH (molar mass 40.00 g/mol). If solid NaOH is available, how would the student prepare this solution? If 2.00 MNaOH is avail- able, how would the student prepare the solution? To help insure three significant figures in the NaOH molarity, to how many sig- nificant figures should the volumes and mass be determined?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:30
When svante arrhenius first proposed his acid-base theory, he was a doctoral candidate. his professors thought his ideas were unfounded. within a decade, the arrhenius theory of acid-base was widely accepted and praised within the scientific world. arrhenius defined acids as compounds having ionizable hydrogen and bases as compounds with ionizable a) barium. b) hydronium. c) hydroxide. d) oxygen.
Answers: 3

Chemistry, 22.06.2019 13:30
If the concentration of phosphate in the cytosol is 2.0 mm and the concentration of phosphate in the surrounding fluid is 0.1 mm, how could the cell increase the concentration of phosphate in the cytosol? a) passive transportb) diffusionc) active transportd) osmosise) facilitated diffusion
Answers: 3

Chemistry, 22.06.2019 16:50
Ajet plane is speeding down the runway during takeoff. air resistance is not negligible. identify the forces on the jet.
Answers: 3

Chemistry, 22.06.2019 23:30
The comparison of the number of atoms in a copper coin the size of a penny with the number of people on earth is made to illustrate which of the following? a. that atoms are indivisible b. that atoms are very small c. that atoms are very large d. that in a copper penny, there is one atom for every person on earth
Answers: 1
You know the right answer?
A student wants to prepare 1.00 L of a 1.00 M solution of NaOH (molar mass 40.00 g/mol). If solid Na...
Questions



Mathematics, 02.12.2021 14:00



Mathematics, 02.12.2021 14:00




Mathematics, 02.12.2021 14:00




English, 02.12.2021 14:00

Mathematics, 02.12.2021 14:00




History, 02.12.2021 14:00

Mathematics, 02.12.2021 14:00

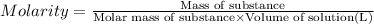
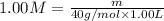
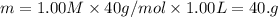
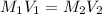 (dilution equation)
(dilution equation)