
Chemistry, 20.03.2020 10:26 cearadenney7067
The rate constant of a certain reaction is known to obey the Arrhenius equation, and to have an activation energy Ea= 50.0 KJ/mol. If the rate constant of this reaction is 0.14M-1 s-1 at -8.0what will the rate constant be at 56.0 ?
Round your answer to significant digits
k= M-1s-1

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 10:20
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3

Chemistry, 22.06.2019 12:30
Which of the following describes a compound? (hint: carbon and oxygen bo a. a piece of pure carbon, containing only carbon atoms b. oxygen gas surrounding a solid piece of carbon c. a substance made of two oxygen atoms for each carbon atom carbon and oxygen atoms mixed without being bonded together
Answers: 1


Chemistry, 22.06.2019 13:30
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1
You know the right answer?
The rate constant of a certain reaction is known to obey the Arrhenius equation, and to have an acti...
Questions

Mathematics, 05.03.2021 16:20

Social Studies, 05.03.2021 16:20

History, 05.03.2021 16:20



Biology, 05.03.2021 16:30

Mathematics, 05.03.2021 16:30

Social Studies, 05.03.2021 16:30



English, 05.03.2021 16:30

Mathematics, 05.03.2021 16:30



Chemistry, 05.03.2021 16:30

Mathematics, 05.03.2021 16:30




Mathematics, 05.03.2021 16:30

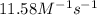
![\ln(\frac{K_{56^oC}}{K_{-8^oC}})=\frac{E_a}{R}[\frac{1}{T_1}-\frac{1}{T_2}]](/tpl/images/0555/9521/c3b12.png)
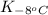 = equilibrium constant at -8°C =
= equilibrium constant at -8°C = 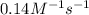
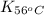 = equilibrium constant at 56°C = ?
= equilibrium constant at 56°C = ?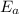 = Activation energy = 50.0 kJ/mol = 50000 J/mol (Conversion factor: 1 kJ = 1000 J)
= Activation energy = 50.0 kJ/mol = 50000 J/mol (Conversion factor: 1 kJ = 1000 J)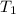 = initial temperature =
= initial temperature = ![-8^oC=[273-8]K=265K](/tpl/images/0555/9521/87944.png)
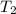 = final temperature =
= final temperature = ![56^oC=[273+56]K=329K](/tpl/images/0555/9521/333d6.png)
![\ln(\frac{K_{56^oC}}{0.14})=\frac{50000J}{8.314J/mol.K}[\frac{1}{265}-\frac{1}{329}]\\\\K_{56^oC}=11.58M^{-1}s^{-1}](/tpl/images/0555/9521/de71d.png)


