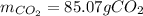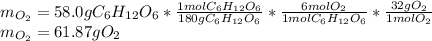Glucose, C 6 H 12 O 6 , is used as an energy source by the human body. The overall reaction in the body is described by the equation C 6 H 12 O 6 ( aq ) + 6 O 2 ( g ) ⟶ 6 CO 2 ( g ) + 6 H 2 O ( l ) Calculate the number of grams of oxygen required to convert 58.0 g of glucose to CO 2 and H 2 O . mass of O 2 : 61.76 g Calculate the number of grams of CO 2 produced.

Answers: 2


Another question on Chemistry

Chemistry, 23.06.2019 00:00
(04.05 hc) analyze the given diagram of the carbon cycle below. part 1: which compound does c represent? part 2: name a process that could release this compound into the air. part 3: explain how the elements that form it are conserved during the carbon cycle. use complete sentences to explain your answer. justify how this compound was created from a recycling of carbon in the carbon cycle. use complete sentences to explain your answer.
Answers: 3


Chemistry, 23.06.2019 03:30
In general metals get as you move from left to right across the periodic table.
Answers: 1

Chemistry, 23.06.2019 06:00
What physical property of gold makes panning a useful way to get gold from streams?
Answers: 2
You know the right answer?
Glucose, C 6 H 12 O 6 , is used as an energy source by the human body. The overall reaction in the b...
Questions

Chemistry, 26.09.2019 22:00



Computers and Technology, 26.09.2019 22:00


Mathematics, 26.09.2019 22:00




Advanced Placement (AP), 26.09.2019 22:00

English, 26.09.2019 22:00