Chemistry, 20.03.2020 05:50 jonystroyer1020
Given that a chlorine-oxygen bond has an enthalpy of 243 kJ/mol , an oxygen-oxygen bond has an enthalpy of 498 kJ/mol , and the standard enthalpy of formation of ClO2 102.5 kJ/mol , calculate the value for the enthalpy of formation per mole of ClO(g). What is the value for the enthalpy of formation per mole of ClO(g)?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 08:30
Which of the following would have less momentum than a 52 kg cheetah running at 10 m/s?
Answers: 2


Chemistry, 22.06.2019 23:00
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1

Chemistry, 23.06.2019 13:30
Type the correct answer in each box. use numerals instead of words. the distance between earth and the moon is about 384,400 kilometers. the distance can be written in scientific notation as a × 10b kilometers, where a = and b = .
Answers: 1
You know the right answer?
Given that a chlorine-oxygen bond has an enthalpy of 243 kJ/mol , an oxygen-oxygen bond has an entha...
Questions


Mathematics, 05.01.2020 13:31


History, 05.01.2020 13:31

Mathematics, 05.01.2020 13:31


Geography, 05.01.2020 13:31


Health, 05.01.2020 13:31

Mathematics, 05.01.2020 13:31

English, 05.01.2020 13:31

Physics, 05.01.2020 13:31


Mathematics, 05.01.2020 13:31



History, 05.01.2020 13:31

Mathematics, 05.01.2020 13:31


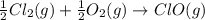
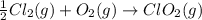 ;
; 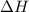 = 102.5 kJ
= 102.5 kJ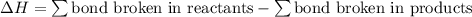
![[(\frac{1}{2})x + 498] - [(2)(243)]](/tpl/images/0555/5141/c814b.png)
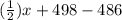
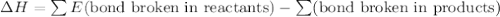
![[(\frac{1}{2})181 + (\frac{1}{2})498] - 243](/tpl/images/0555/5141/97422.png)