
Chemistry, 20.03.2020 01:40 churchlady114p2met3
Write equations that show the processes that describe the first, second, and third ionization energies for a gaseous gadolinium atom. Express your answers as chemical equations separated by commas. Identify all of the phases in your answer.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 22.06.2019 23:00
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1

Chemistry, 23.06.2019 06:00
What are the coefficients to balance the following equation? ba+br2=babr2
Answers: 2

Chemistry, 23.06.2019 08:40
Which statement is true according to the kinetic theory? a. molecules of different gases with the same mass and temperature always have the same average density. b. molecules of different gases with the same mass and temperature always have the same average volume. c. molecules of different gases with the same mass and temperature always have the same pressure. d. molecules of different gases with the same mass and temperature always have the same molecular mass. e. molecules of different gases with the same mass and temperature always have the same average kinetic energy.
Answers: 1
You know the right answer?
Write equations that show the processes that describe the first, second, and third ionization energi...
Questions





Mathematics, 06.05.2020 21:00




Mathematics, 06.05.2020 21:00



Mathematics, 06.05.2020 21:00

Mathematics, 06.05.2020 21:00


Mathematics, 06.05.2020 21:00


History, 06.05.2020 21:00


Mathematics, 06.05.2020 21:00

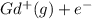 ,
, 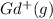 →
→ 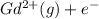 ,
, 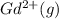 →
→ 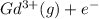
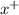 ion. The second ionization energy is always higher than the first:
ion. The second ionization energy is always higher than the first: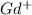 (g) →
(g) → 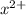 ion:
ion: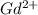 (g) →
(g) → 

