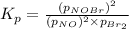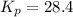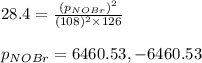Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:30
What is the relationship of air masses and the temperature of oceans?
Answers: 1

Chemistry, 22.06.2019 10:30
Aglow stick contains a glass vial with chemicals. when the glow stick is bent, the vial breaks and the chemicals react to produce a glow. a science student observes that a glow stick kept in the freezer glows for a longer duration than a glow stick kept at room temperature. what conclusion can be drawn based on the observation? be sure to note the outcome and test variables in the conclusion.
Answers: 1

Chemistry, 22.06.2019 11:00
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1

Chemistry, 23.06.2019 01:00
Reactions in cells take place at about a. 40°c b. 0° c. 100°c d. 60°c
Answers: 1
You know the right answer?
Consider the reaction: 2 NO(g) + Br2(g) ∆ 2 NOBr(g) Kp = In a reaction mixture at equilibrium, the p...
Questions



Social Studies, 04.08.2019 06:30


Mathematics, 04.08.2019 06:30

History, 04.08.2019 06:30

Mathematics, 04.08.2019 06:30

Business, 04.08.2019 06:30

Mathematics, 04.08.2019 06:30

Social Studies, 04.08.2019 06:30

History, 04.08.2019 06:30

Mathematics, 04.08.2019 06:30

Business, 04.08.2019 06:30


History, 04.08.2019 06:30

Social Studies, 04.08.2019 06:30

History, 04.08.2019 06:30

Mathematics, 04.08.2019 06:30


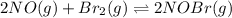
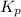 for above equation follows:
for above equation follows: