
Chemistry, 20.03.2020 00:06 buddyclayjohnson
A mixture of methane (CH4) and ethane (C2H6) is stored in a container at 294 mm Hg. The gases are burned in air to form CO2 and H2O. If the pressure of CO2 is 351 mm Hg measured at the same temperature and volume as the original mixture, calculate the mole fraction of the gases.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 15:00
Answer explain why it is not possible to deduce a complete order of reactivity.
Answers: 3

Chemistry, 23.06.2019 04:00
If you are told to get 100 ml of stock solution to use to prepare smaller size sample for an experiment, which piece of glassware would you use?
Answers: 3

Chemistry, 23.06.2019 08:00
Ineed this awnser fast select the correct answer. this chemical equation represents the burning of methane, but the equation is incomplete. what is the missing coefficient in both the reactants and the products? ch4 + → co2 + a. 0 b. 1c. 2d. 3 e. 4
Answers: 3

Chemistry, 23.06.2019 08:00
Pl what kind of reaction is this? nahco3 + h2o → co2 + naoh + h2o -composition -decomposition -single replacement -double replacement im leaning more toward single replacement. if im wrong can you explain whyy?
Answers: 1
You know the right answer?
A mixture of methane (CH4) and ethane (C2H6) is stored in a container at 294 mm Hg. The gases are bu...
Questions

Mathematics, 27.06.2019 19:20

Mathematics, 27.06.2019 19:20

History, 27.06.2019 19:20


Mathematics, 27.06.2019 19:20


English, 27.06.2019 19:20



Mathematics, 27.06.2019 19:20

English, 27.06.2019 19:20

Mathematics, 27.06.2019 19:20

English, 27.06.2019 19:20

Physics, 27.06.2019 19:20

Mathematics, 27.06.2019 19:20



Mathematics, 27.06.2019 19:20


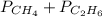 = 294 mm Hg
= 294 mm Hg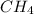 will yield 1 mole of
will yield 1 mole of 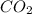 whereas 1 mole of
whereas 1 mole of 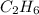 will yield 2 moles of
will yield 2 moles of 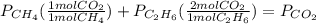
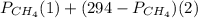 = 351 mm Hg
= 351 mm Hg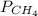 = 2(294) - 351
= 2(294) - 351
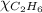 = 1 - 0.806
= 1 - 0.806

