
For the reaction CO2(g) + H2O(ℓ) + CaCl2(aq) ←→ CaCO3(s) + HCl(aq) K = 4.8 the concentrations of CO2, H2O, CaCl2 and HCl are 0.377 M, 55.4 M, 0.652 M and 30.2 M, respectively. What will happen in order for the system to reach equilibrium? 1. The reaction will shift to the right. 2. Nothing will occur. 3. The reaction will shift to the left. 4. Not enough information is given.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:30
Asample of the male sex hormone testosterone, c19h28o2, contains 3.88×10^21 atoms of hydrogen.(a) how many atoms of carbon does it contain? (b) how many molecules of testosterone does it contain? (c) how many moles of testosterone does it contain? (d) what is the mass of this sample in grams?
Answers: 1

Chemistry, 21.06.2019 18:50
Which term refers to a property that depends only on the amount of a substance? ©@
Answers: 2

Chemistry, 22.06.2019 10:10
Stage in which a typical star has completely stopped fusion
Answers: 1

You know the right answer?
For the reaction CO2(g) + H2O(ℓ) + CaCl2(aq) ←→ CaCO3(s) + HCl(aq) K = 4.8 the concentrations of CO2...
Questions

Social Studies, 03.12.2021 01:00

Computers and Technology, 03.12.2021 01:00

Social Studies, 03.12.2021 01:00


Computers and Technology, 03.12.2021 01:00

English, 03.12.2021 01:00


Mathematics, 03.12.2021 01:00

Chemistry, 03.12.2021 01:00





Social Studies, 03.12.2021 01:00

English, 03.12.2021 01:00

Computers and Technology, 03.12.2021 01:00


Mathematics, 03.12.2021 01:00

Computers and Technology, 03.12.2021 01:00

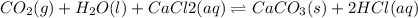
![K=\frac{[HCl]^2}{[CaCl_2]}](/tpl/images/0554/8954/74ccf.png)
![Q=\frac{[HCl]^2}{[CaCl_2]}=\frac{ 30.2^2 }{0.652 }=1398.8](/tpl/images/0554/8954/f4b79.png)


