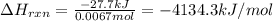Chemistry, 19.03.2020 21:25 discolldiane7351
A sample of hexane (C6H14) has a mass of 0.580 g. The sample is burned in a bomb calorimeter that has a mass of 1.900 kg and a specific heat of 3.21 J/giK. What amount of heat is produced during the combustion of hexane if the temperature of the calorimeter increases by 4.542 K? Use .

Answers: 1


Another question on Chemistry

Chemistry, 23.06.2019 04:20
The graph shows one consequence of urban sprawl. how did urban sprawl contribute to the change in biodiversity
Answers: 2

Chemistry, 23.06.2019 05:50
What are the coefficients to balance the following equation? ba+br=babr2
Answers: 1


Chemistry, 23.06.2019 10:30
Most ionic compouds are crystalline solids at room temperature. true falseionic compounds are electrically neutral. true falseionic compounds generally have low melting points. true falsewhen melted, ionic compounds do not conduct electricity. true falsethe electrostatic attraction between an anion and a cation is an ionic bond. true false
Answers: 1
You know the right answer?
A sample of hexane (C6H14) has a mass of 0.580 g. The sample is burned in a bomb calorimeter that ha...
Questions

Spanish, 09.10.2019 04:30

Health, 09.10.2019 04:30

Mathematics, 09.10.2019 04:30

Health, 09.10.2019 04:30

Social Studies, 09.10.2019 04:30



Social Studies, 09.10.2019 04:30


Biology, 09.10.2019 04:30

Mathematics, 09.10.2019 04:30

Mathematics, 09.10.2019 04:30

Mathematics, 09.10.2019 04:30


Mathematics, 09.10.2019 04:30

English, 09.10.2019 04:30


Mathematics, 09.10.2019 04:30

Social Studies, 09.10.2019 04:30

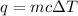
 = change in temperature = 4.542 K
= change in temperature = 4.542 K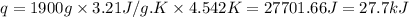
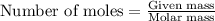
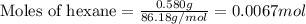
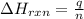
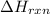 = enthalpy change of the reaction
= enthalpy change of the reaction