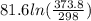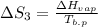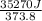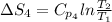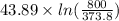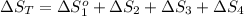The standard entropy of liquid methanol at 298K is 126.8 J/K-mol and its heat capacity is 81.6 J/K-mol. Methanol boils at 337K with an enthalpy of vaporization of 35.270 kJ/mol at that temperature. The heat capacity of the vapor is 43.9 J/K-mol.__Calculate the entropy of one mole of methanol vapor at 800 K.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1

Chemistry, 22.06.2019 15:00
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1

Chemistry, 23.06.2019 02:00
Now look at the segment of the graph between the two data points marked with black squares. describe how the boiling point and melting point plots behave between these points. be as specific as possible.
Answers: 1

Chemistry, 23.06.2019 06:50
Organisms are classified as producer or consumers according to the way they ?
Answers: 1
You know the right answer?
The standard entropy of liquid methanol at 298K is 126.8 J/K-mol and its heat capacity is 81.6 J/K-m...
Questions

Computers and Technology, 25.02.2021 21:30

Arts, 25.02.2021 21:30



Mathematics, 25.02.2021 21:30






English, 25.02.2021 21:30



Spanish, 25.02.2021 21:30



Mathematics, 25.02.2021 21:30

Mathematics, 25.02.2021 21:30

Mathematics, 25.02.2021 21:30

Mathematics, 25.02.2021 21:30

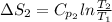 J/K mol
J/K mol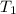 = 298 K,
= 298 K, 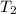 = 373.8 K
= 373.8 K