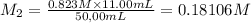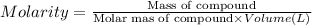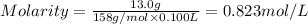Chemistry, 19.03.2020 20:56 Uhmjujiooo45701
By pipet, 11.00 mL of a 0.823 MM stock solution of potassium permanganate (KMnO4) was transferred to a 50.00-mL volumetric flask and diluted to the calibration mark. Determine the molarity of the resulting solution. A stock solution of potassium permanganate (KMnO4) was prepared by dissolving 13.0g KMnO4 with DI H2O in a 100.00-mL volumetric flask and diluting to the calibration mark. Determine the molarity of the solution Molarity= O.822 M

Answers: 2


Another question on Chemistry


Chemistry, 23.06.2019 07:00
Under what conditions will a gas be most likely to exhibit the ideal gas properties predicted by the ideal gas law? 1)high pressures and high temperature, because particles are forced closer together with higher kinetic energy, so intermolecular forces of attraction are weaker 2)high pressure and low temperature, because particles are forced closer together and moving slower, so the volume of the particles is less significant 3) low pressure and high temperature, because particles are spread farther apart and moving faster, so the intermolecular forces of attraction are weaker 4)low pressure and low temperature, because particles are spread farther apart with lower kinetic energy, so the volume of the particles is less significant
Answers: 2


Chemistry, 23.06.2019 15:30
Which answer below correctly identifies the type of change and the explanation when magnesium comes into contact with hydrochloric acid
Answers: 1
You know the right answer?
By pipet, 11.00 mL of a 0.823 MM stock solution of potassium permanganate (KMnO4) was transferred to...
Questions





Mathematics, 30.05.2020 09:00


Mathematics, 30.05.2020 09:00

History, 30.05.2020 09:00

Mathematics, 30.05.2020 09:00


Mathematics, 30.05.2020 09:00



History, 30.05.2020 09:00



Spanish, 30.05.2020 09:00

Mathematics, 30.05.2020 09:00


Mathematics, 30.05.2020 09:00

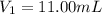
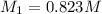
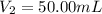
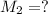
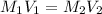 ( dilution )
( dilution )