
Chemistry, 19.03.2020 08:58 jetblackcap
Phosphorus pentachloride decomposes according to the chemical equation PCl 5 ( g ) − ⇀ ↽ − PCl 3 ( g ) + Cl 2 ( g ) K c = 1.80 at 250 ∘ C A 0.1846 mol sample of PCl 5 ( g ) is injected into an empty 2.55 L reaction vessel held at 250 ∘ C. Calculate the concentrations of PCl 5 ( g ) and PCl 3 ( g ) at equilibrium.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:00
According to the tide table below what time of day will the highest tide occur?
Answers: 1

Chemistry, 23.06.2019 01:00
An unsaturated hydrocarbon is a hydrogen-carbon compound with a. a network solid structure b. single bonds c. single bonds in a branched-chain structure d. double or triple bonds
Answers: 1

Chemistry, 23.06.2019 07:00
What is the difference between covalent bonds and ionic bonds? covalent bonds involve the sharing of electrons between atoms; ionic bonds involve the electrical attraction between charged atoms. covalent bonds involve the transfer of electrons between charged atoms; ionic bonds involve the sharing of electrons between atoms. covalent bonds involve the sharing of pairs of electrons between atoms; ionic bonds involve the sharing of single electrons between atoms. covalent bonds involve the sharing of electrons between atoms; ionic bonds involve the sharing of protons between charged atoms.
Answers: 1

Chemistry, 23.06.2019 11:00
Which of the following reactions represents an exothermic reaction? nh3(g) + 12.0 kcal ½n2(g) + 3/2 h2(g) ch4 + 2o2 co2 + 2h2o + 212,800 cal c + 2s cs2, h = 27,550 cal c(graphite) c(diamond), h = 0.45 kcal 2h2o 2h2 + o2, h = +58 kcal
Answers: 1
You know the right answer?
Phosphorus pentachloride decomposes according to the chemical equation PCl 5 ( g ) − ⇀ ↽ − PCl 3 ( g...
Questions

History, 07.12.2020 23:50


Mathematics, 07.12.2020 23:50

Mathematics, 07.12.2020 23:50


Mathematics, 07.12.2020 23:50







Mathematics, 07.12.2020 23:50

English, 07.12.2020 23:50


Mathematics, 07.12.2020 23:50




![[PCl_3]_{eq}=0.0697M\\](/tpl/images/0553/8714/e4a7f.png)
![[PCl_5]_{eq}=0.00269M](/tpl/images/0553/8714/6813e.png)
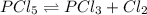
![[PCl_5]_0=\frac{0.1846mol}{2.55L}=0.0724M](/tpl/images/0553/8714/c9006.png)
![Kc=\frac{[Cl_2]_{eq}[PCl_3]_{eq}}{[PCl_5]_{eq}}](/tpl/images/0553/8714/38534.png)
 due to the reaction extent, it becomes:
due to the reaction extent, it becomes: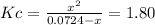
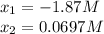
![[PCl_3]_{eq}=x=0.0697M\\](/tpl/images/0553/8714/b7f0d.png)
![[PCl_5]_{eq}=0.0724M-x=0.0724M-0.0697M=0.00269M](/tpl/images/0553/8714/fc102.png)


