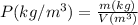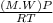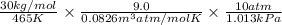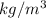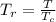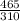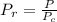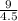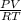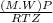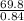Chemistry, 19.03.2020 07:58 kelseiroll9759
A certain gas has a molecular weight of 30.0, a critical temperature of 310 K, and a critical pressure of 4.5 MPa. Calculate the density in kg / m 3 kg/m3 of this gas at 465 K and 9.0 MPa (a) if the gas is ideal and (b) if the gas obeys the law of corresponding states

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Match the following items. 1. high-intensity bundle of energy being emitted from some decaying nuclei gamma ray 2. particle radiating from the nucleus of some atoms beta particle 3. negative particle identical to an electron but radiating from a decaying nucleus alpha particle
Answers: 1

Chemistry, 22.06.2019 13:00
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2

Chemistry, 22.06.2019 14:00
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1
You know the right answer?
A certain gas has a molecular weight of 30.0, a critical temperature of 310 K, and a critical pressu...
Questions




Arts, 14.07.2019 18:30



Physics, 14.07.2019 18:30

Geography, 14.07.2019 18:30


History, 14.07.2019 18:30



World Languages, 14.07.2019 18:30


History, 14.07.2019 18:30