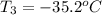Chemistry, 19.03.2020 07:42 kcceff4085
A food product is being frozen in a system capable of removing 6000 kJ of thermal energy. The product has a specifi c heat of 4 kJ/(kg C) above the freezing temperature of 2 C, the latent heat of fusion equals 275 kJ/kg, and the frozen product has a specifi c heat of 2.5 kJ/(kg C) below 2 C. If 10 kg of product enters the system at 10C, determine the exit temperature of the product..

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

Chemistry, 22.06.2019 12:00
Why are people not able to skip a dive to the deepest part of the ocean
Answers: 1

Chemistry, 22.06.2019 13:30
If the concentration of phosphate in the cytosol is 2.0 mm and the concentration of phosphate in the surrounding fluid is 0.1 mm, how could the cell increase the concentration of phosphate in the cytosol? a) passive transportb) diffusionc) active transportd) osmosise) facilitated diffusion
Answers: 3
You know the right answer?
A food product is being frozen in a system capable of removing 6000 kJ of thermal energy. The produc...
Questions

Mathematics, 05.05.2020 05:16











Computers and Technology, 05.05.2020 05:16

Mathematics, 05.05.2020 05:16

History, 05.05.2020 05:16

Biology, 05.05.2020 05:16

Mathematics, 05.05.2020 05:16



Computers and Technology, 05.05.2020 05:16

Physics, 05.05.2020 05:16

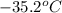
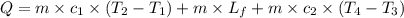
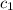 = specific heat of liquid =
= specific heat of liquid = 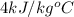
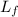 = latent heat of fusion =
= latent heat of fusion = 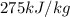
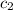 = specific heat of frozen =
= specific heat of frozen = 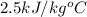
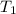 = initial temperature of liquid =
= initial temperature of liquid = 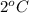
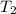 = final temperature of liquid =
= final temperature of liquid = 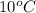
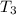 = initial temperature of frozen = ?
= initial temperature of frozen = ?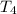 = final temperature of frozen =
= final temperature of frozen = ![6000kJ=[15kg\times 4kJ/kg^oC\times (10-2)^oC]+[15kg\times 275kJ/kg]+[15kg\times 2.5kJ/kg^oC\times (2-T_3)^oC]](/tpl/images/0553/7186/e8cbe.png)