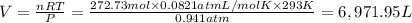Chemistry, 19.03.2020 01:58 tristasbarker03
Calculate the volume of carbon dioxide at 20.0°C and 0.941 atm produced from the complete combustion of 4.00 kg of methane. Compare your result with the volume of CO2 produced from the complete combustion of 4.00 kg of propane (C3H8).

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:00
In the 1800s, one of the statements in john dalton's atomic theory was that atoms are indivisible. later experimental evidence led to the discovery of subatomic particles such as neutrons, electrons, and protons. what happened to the indivisible atom part of dalton's atomic theory, and why?
Answers: 3

Chemistry, 22.06.2019 19:30
To calculate percent by mass, use the equation below: calculate the percent by mass of each element. %n = % %h = % %o = %
Answers: 3

Chemistry, 23.06.2019 12:20
Amatch has about 21 milligrams of red phosphorus coating the tip. how many atoms of phosphorus is this?
Answers: 1

Chemistry, 23.06.2019 18:10
A1 forms when an acid is neutralized by a base. 1. salts can be neutral, or in solutions. salts of 2. strong acid–strong base reactions produce solutions with 3. water. salts formed from the neutralization of weak acids or weak 4. bases water. they produce solutions that are acidic or . basic. for example, the ph of a solution at the equivalence point is . greater than for a acid titration. solutions that resist changes in ph are called solutions. the buffer is the amount of acid or base that can be added to a buffer without changing the ph greatly.
Answers: 1
You know the right answer?
Calculate the volume of carbon dioxide at 20.0°C and 0.941 atm produced from the complete combustion...
Questions


History, 20.10.2020 20:01


Mathematics, 20.10.2020 20:01



Mathematics, 20.10.2020 20:01



Mathematics, 20.10.2020 20:01






Social Studies, 20.10.2020 20:01

History, 20.10.2020 20:01


Biology, 20.10.2020 20:01

Mathematics, 20.10.2020 20:01

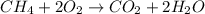
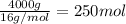
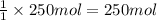 of carbon dioxide gas
of carbon dioxide gas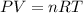 (Ideal gas equation)
(Ideal gas equation)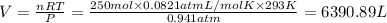
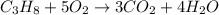
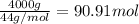
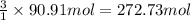 of carbon dioxide gas
of carbon dioxide gas