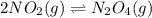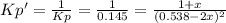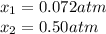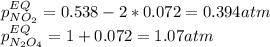Chemistry, 18.03.2020 21:59 briweaver9993
• Consider the reaction N2O4 (g) ⇌ 2 NO2 (g). At equilibrium, a 2.00-L reaction vessel contains NO2 at a pressure of 0.269 atm and N2O4 at a pressure of 0.500 atm. The reaction vessel is then compressed to 1.00 L. What will be the pressures of NO2 and N2O4 once equilibrium is re-established?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:30
You are performing an experiment in a lab to attempt a new method of producing pure elements from compounds. the only problem is that you do not know what element will form. by your previous calculations you know that you will have 6.3 moles of product. when it is complete, you weigh it and determine you have 604.4 grams. what element have you produced?
Answers: 1

Chemistry, 22.06.2019 06:30
How many moles of carbon dioxide will form if 2.5 moles of c3h8 is burned
Answers: 1

Chemistry, 22.06.2019 09:00
What term is missing from the central region that describes hypotheses, theories, and laws? popular predictable mathematical falsifiable
Answers: 2

You know the right answer?
• Consider the reaction N2O4 (g) ⇌ 2 NO2 (g). At equilibrium, a 2.00-L reaction vessel contains NO2...
Questions


History, 27.01.2020 18:31


Social Studies, 27.01.2020 18:31


Mathematics, 27.01.2020 18:31

Social Studies, 27.01.2020 18:31





History, 27.01.2020 18:31



Mathematics, 27.01.2020 18:31


Arts, 27.01.2020 18:31

Biology, 27.01.2020 18:31


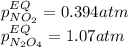
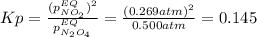
 is now present in order to take the reaction again to the equilibrium. Besides, the reaction changes as the products have more moles as:
is now present in order to take the reaction again to the equilibrium. Besides, the reaction changes as the products have more moles as: