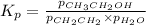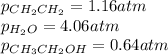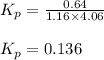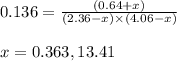Chemistry, 18.03.2020 22:08 kellinvagneur
While ethanol is produced naturally by fermentation, e. g. in beer- and wine-making, industrially it is synthesized by reacting ethylene with water vapor at elevated temperatures. A chemical engineer studying this reaction fills a flask with of ethylene gas and of water vapor. When the mixture has come to equilibrium she determines that it contains of ethylene gas and of water vapor. The engineer then adds another of ethylene, and allows the mixture to come to equilibrium again. Calculate the pressure of ethanol after equilibrium is reached the second time. Round your answer to significant digits.

Answers: 2


Another question on Chemistry


Chemistry, 21.06.2019 20:30
When curium-242 is bombarded with an alpha particle, two products are formed, one of which is a nudge on. what is the other product
Answers: 3

Chemistry, 22.06.2019 04:30
Use the periodic table to determine the electron configuration of dysprosium (dy) and americium (am) in noble-gas notation.
Answers: 1

Chemistry, 22.06.2019 12:00
What term is applied to a scientist who studies ancient life, including animal and plant fossils a. anthropologist b. dendroclimatologist c. geophysicist d. paleontologist
Answers: 2
You know the right answer?
While ethanol is produced naturally by fermentation, e. g. in beer- and wine-making, industrially it...
Questions

English, 09.04.2021 20:10

Mathematics, 09.04.2021 20:10

English, 09.04.2021 20:10




Mathematics, 09.04.2021 20:10

Mathematics, 09.04.2021 20:10

Mathematics, 09.04.2021 20:10

Physics, 09.04.2021 20:10


Mathematics, 09.04.2021 20:10

Chemistry, 09.04.2021 20:10




Mathematics, 09.04.2021 20:10

Mathematics, 09.04.2021 20:10


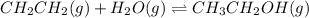
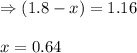
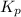 for above equation follows:
for above equation follows: