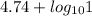Acetic acid has a Ka of 1.8*10^-5. Three acetic acid/ acetate buffer solutions, A, B, and C, wer made using varying concentrations: 1. [acetic acid] ten times greater than [acetate] 2. [acetate] ten times greater than [acetic acid] 3. [acetate] = [acetic acid] Match each buffer to the expected pH pH = 3.74 pH= 4.74 pH = 5.74

Answers: 1


Another question on Chemistry



Chemistry, 22.06.2019 10:30
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2

Chemistry, 22.06.2019 15:00
Which of the following is the correct formula for copper (i) sulfate trihydrate? cuso4 · 3h2o cuso4(h2o)3 cu2so4(h2o)3 cu2so4 · 3h2o
Answers: 1
You know the right answer?
Acetic acid has a Ka of 1.8*10^-5. Three acetic acid/ acetate buffer solutions, A, B, and C, wer mad...
Questions

English, 26.03.2021 20:50




Mathematics, 26.03.2021 20:50



Mathematics, 26.03.2021 20:50

Mathematics, 26.03.2021 20:50


Mathematics, 26.03.2021 20:50

Mathematics, 26.03.2021 20:50

French, 26.03.2021 20:50




English, 26.03.2021 20:50


Chemistry, 26.03.2021 20:50

 for acetic acid is
for acetic acid is  . And, its
. And, its  value will be calculated as follows.
value will be calculated as follows.
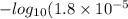
![pK_{a} + log \frac{[Salt]}{[Acid]}](/tpl/images/0550/7017/81f72.png)
![\frac{[\text{Acetate}]}{[\text{Acetic acid}]} = \frac{1}{10}](/tpl/images/0550/7017/c529e.png)
![pK_{a} + log \frac{[Acetate]}{[\text{Acetic Acid}]}](/tpl/images/0550/7017/a17de.png)

![\frac{[Acetate]}{\text{Acetic acid}}](/tpl/images/0550/7017/7d291.png) =
= 
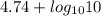
 = 1
= 1