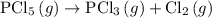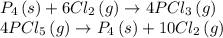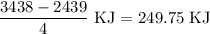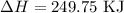Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 20:10
What would happen to a volleyball left outside in the winter? o o o o a. it would expand. b. it would lose air. c. it would shrink. d. it would explode.
Answers: 2

Chemistry, 23.06.2019 05:00
What is dhmo? hint: you find it everywhere something is wet..
Answers: 1

Chemistry, 23.06.2019 09:00
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate according to the following equation:
Answers: 2

Chemistry, 23.06.2019 09:30
Where are the noble gases located in the periodic table? a. in the center b. on the left side c. in the upper right corner d. on the far right side
Answers: 1
You know the right answer?
Find the ΔH for the reaction below, given the following reactions and subsequent ΔH values: PCl5(g)...
Questions

Computers and Technology, 06.12.2019 19:31





English, 06.12.2019 19:31












SAT, 06.12.2019 19:31

Social Studies, 06.12.2019 19:31

Social Studies, 06.12.2019 19:31

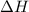 for the desired reaction will be 249.75 KJ.
for the desired reaction will be 249.75 KJ.