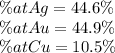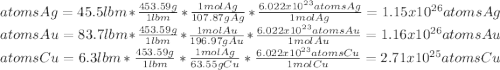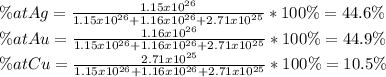Chemistry, 17.03.2020 06:08 elnkun98owvaa6
What is the composition, in atom percent, of an alloy that contains a) 45.5 lbm of silver, b) 83.7 lbm of gold, and c) 6.3 lbm of Cu? The atomic weights for silver, gold, and copper are, respectively, 107.87, 196.97, and 63.55 g/mol.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Which of these would be caused by a chemical change? a) the formation of lava. b) sedimantary rock layering over time. c) metamorphic rock forming from igneous. d) metamorphic rock eroding to form sedimentary rock.
Answers: 3

Chemistry, 22.06.2019 18:10
Given the following at 25c calculate delta hf for hcn (g) at 25c. 2nh3 (g) +3o2 (g) + 2ch4 (g) > 2hcn (g) + 6h2o (g) delta h rxn= -870.8 kj. delta hf=-80.3 kj/mol for nh3 (g), -74.6 kj/mol for ch4, and -241.8 kj/mol for h2o (g)
Answers: 1

Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1

Chemistry, 22.06.2019 21:00
Which of the following is a physical property flammability heat of combustion solubility and toxicity
Answers: 1
You know the right answer?
What is the composition, in atom percent, of an alloy that contains a) 45.5 lbm of silver, b) 83.7 l...
Questions











Geography, 27.09.2019 16:30

Social Studies, 27.09.2019 16:30



Chemistry, 27.09.2019 16:30

Biology, 27.09.2019 16:30



Mathematics, 27.09.2019 16:30

Health, 27.09.2019 16:30