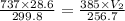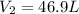A weather balloon is inflated to a volume of 28.6 L at a pressure of 737 mmHg and a temperature of 26.8 ∘C. The balloon rises in the atmosphere to an altitude where the pressure is 385 mmHg and the temperature is -16.3 ∘C. Assuming the balloon can freely expand, calculate the volume of the balloon at this altitude.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:30
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2

Chemistry, 22.06.2019 10:10
What shape would a molecule with two bound groups and two lone pairs have?
Answers: 1


Chemistry, 22.06.2019 23:00
What is the energy in joules of a mole of photons associated with visible light of wavelength 486 nm?
Answers: 3
You know the right answer?
A weather balloon is inflated to a volume of 28.6 L at a pressure of 737 mmHg and a temperature of 2...
Questions


Chemistry, 17.11.2020 14:00




Social Studies, 17.11.2020 14:00


Mathematics, 17.11.2020 14:00

English, 17.11.2020 14:00


Biology, 17.11.2020 14:00



English, 17.11.2020 14:00




Mathematics, 17.11.2020 14:00

Social Studies, 17.11.2020 14:00

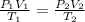
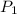 = initial pressure of gas = 737 mm Hg
= initial pressure of gas = 737 mm Hg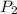 = final pressure of gas = 385 mm Hg
= final pressure of gas = 385 mm Hg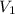 = initial volume of gas = 28.6 L
= initial volume of gas = 28.6 L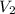 = final volume of gas = ?
= final volume of gas = ?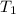 = initial temperature of gas =
= initial temperature of gas = 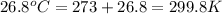
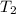 = final temperature of gas =
= final temperature of gas =