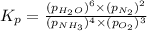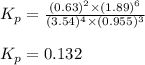Chemistry, 16.03.2020 22:22 fordalolz1553
Problem Page Question Ammonia has been studied as an alternative "clean" fuel for internal combustion engines, since its reaction with oxygen produces only nitrogen and water vapor, and in the liquid form it is easily transported. An industrial chemist studying this reaction fills a flask with of ammonia gas and of oxygen gas, and when the mixture has come to equilibrium measures the partial pressure of water vapor to be . Calculate the pressure equilibrium constant for the combustion of ammonia at the final temperature of the mixture. Round your answer to significant digits. Clears your work. Undoes your last action. Provides information about entering answers.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 13:00
What happens to the average kinetic energy of a gas when the particles of the gas collide against each other at a constant temperature and volume? explain your answer.
Answers: 3


Chemistry, 22.06.2019 15:30
Which suspect most likely committed the robbery and how do you know
Answers: 2

You know the right answer?
Problem Page Question Ammonia has been studied as an alternative "clean" fuel for internal combustio...
Questions

Advanced Placement (AP), 18.12.2020 02:40

Mathematics, 18.12.2020 02:40



Arts, 18.12.2020 02:50


Chemistry, 18.12.2020 02:50

Mathematics, 18.12.2020 02:50

Mathematics, 18.12.2020 02:50





Mathematics, 18.12.2020 02:50

Mathematics, 18.12.2020 02:50

Chemistry, 18.12.2020 02:50

Biology, 18.12.2020 02:50


Mathematics, 18.12.2020 02:50

Mathematics, 18.12.2020 02:50

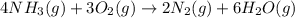
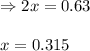
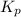 for above equation follows:
for above equation follows: