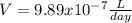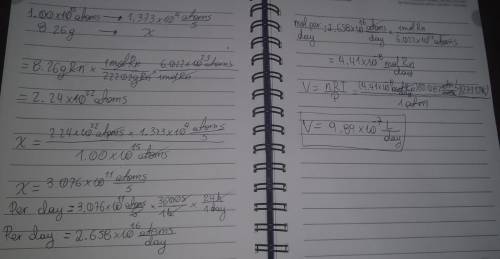Chemistry, 16.03.2020 22:19 dbn4everloved8
Radon (Rn) is the heaviest and the only radioactive member of Group 8A(18), the noble gases. It is a product of the disintegration of heavier radioactive nuclei found in minute concentrations in many common rocks used for building and construction. In recent years, health concerns about the cancers caused from inhaled residential radon have grown. If 1.00 × 1015 atoms of radium (Ra) produce an average of 1.373 × 104 atoms of Rn per second, how many liters of Rn, measured at STP, are produced per day by 8.26 g of Ra?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:30
Which of the following represents the ester functional group? a. -coo- b. -cho c. -cooh d. c=o
Answers: 1

Chemistry, 22.06.2019 14:40
Choose an equation that represents an enzyme-catalyzed reaction. (a) enzyme + substrate → enzyme-substrate complex (b) enzyme + substrate ←−→ enzyme + products (c) enzyme + substrate ←−→ enzyme-substrate complex → enzyme + products (d) enzyme + substrate ←−→ enzyme-substrate complex → enzyme-substrate complex + products
Answers: 2

Chemistry, 22.06.2019 15:00
According to the diagram, what sources contribute to the phosphorus found in soil? according to the diagram, phosphorus found in soil contributes phosphorus to what other sources?
Answers: 1

Chemistry, 22.06.2019 16:40
Let the ed50 of a recreational drug be defined as the amount required for 50% of a test group to feel high or get a buzz. if the ed50 value of ethanol is 470 mg/kg body mass, what dose would a 70 kg party goer need to quickly consume in order to have a 50% chance of getting a buzz? 235 mg 470 mg 32,900 mg 35,000,000 mg
Answers: 3
You know the right answer?
Radon (Rn) is the heaviest and the only radioactive member of Group 8A(18), the noble gases. It is a...
Questions


Social Studies, 12.10.2019 21:10

Mathematics, 12.10.2019 21:10


Mathematics, 12.10.2019 21:10

Chemistry, 12.10.2019 21:10




History, 12.10.2019 21:10

Mathematics, 12.10.2019 21:10


Mathematics, 12.10.2019 21:10






Mathematics, 12.10.2019 21:10

Mathematics, 12.10.2019 21:10