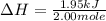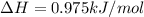Chemistry, 16.03.2020 21:27 bremarie102013
A total of 2.00 mol of a compound is allowed to react with water in a foam coffee cup and the reaction produces 126 gg of solution. The reaction caused the temperature of the solution to rise from 21.00 to 24.70 ∘C∘C. What is the enthalpy of this reaction? Assume that no heat is lost to the surroundings or to the coffee cup itself and that the specific heat of the solution is the same as that of pure water.

Answers: 1


Another question on Chemistry



Chemistry, 22.06.2019 12:00
Under normal conditions, describe how increasing the temperatures effects the solubility of a typical salt
Answers: 1

Chemistry, 23.06.2019 01:30
What happens to the concentration of hydronium ions as the ph of a solution increases? a. hydronium ion concentration stays the same b. hydronium ion concentration decreases c. hydronium ion concentration increases
Answers: 1
You know the right answer?
A total of 2.00 mol of a compound is allowed to react with water in a foam coffee cup and the reacti...
Questions

Biology, 07.07.2019 14:30

Mathematics, 07.07.2019 14:30

Mathematics, 07.07.2019 14:30







History, 07.07.2019 14:30

History, 07.07.2019 14:30

Mathematics, 07.07.2019 14:30

Biology, 07.07.2019 14:30

History, 07.07.2019 14:30



Mathematics, 07.07.2019 14:30

Chemistry, 07.07.2019 14:30


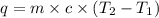
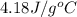
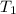 = initial temperature =
= initial temperature = 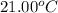
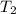 = final temperature =
= final temperature = 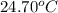
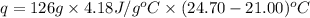
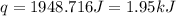
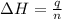
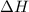 = enthalpy change = ?
= enthalpy change = ?