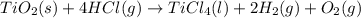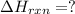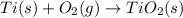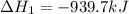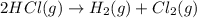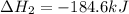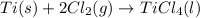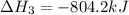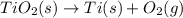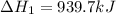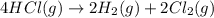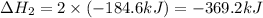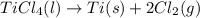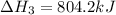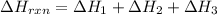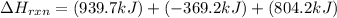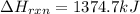Chemistry, 16.03.2020 18:50 kyleighott
Determine the heat of reaction for the process TiO2(s) + 4HCl(g) TiCl4(l) + 2H2(g) + O2(g) using the information given below: Ti(s) + O2(g) TiO2(s) H° = −939.7 kJ 2HCl(g) H2(g) + Cl2(g) H° = −184.6 kJ Ti(s) + 2Cl2(g) TiCl4(l) H° = −804.2 kJ

Answers: 1


Another question on Chemistry

Chemistry, 20.06.2019 18:04
What is the molarity of a solution that is prepared by adding 57.1 g of toluene (c 7 h 8 ) (density = 0.867 g/ml) to a 250 ml volumetric flask, and then filling to the mark with benzene (c 6 h 6 ) (density = 0.876 g/ml)?
Answers: 1

Chemistry, 22.06.2019 00:30
Elements that do not have full outer electron shells will donate, share, or take electrons from other atoms. choose the items that have the correct binary ionic formula.
Answers: 2


You know the right answer?
Determine the heat of reaction for the process TiO2(s) + 4HCl(g) TiCl4(l) + 2H2(g) + O2(g) using the...
Questions

English, 09.11.2019 02:31

SAT, 09.11.2019 02:31

History, 09.11.2019 02:31

History, 09.11.2019 02:31

Mathematics, 09.11.2019 02:31

Health, 09.11.2019 02:31


Mathematics, 09.11.2019 02:31

Social Studies, 09.11.2019 02:31

History, 09.11.2019 02:31

Computers and Technology, 09.11.2019 02:31

Mathematics, 09.11.2019 02:31


Mathematics, 09.11.2019 02:31



Social Studies, 09.11.2019 02:31

Health, 09.11.2019 02:31