
Chemistry, 14.03.2020 06:55 rosemarybooker
The following reaction is spontaneous as written when the components are in their standard states:
3 Zn(s) +2 Cr3+(aq) →3 Zn2+(aq) +2 Cr(s)
If the [Zn2+] is 4 molL−1, determine the value of [Cr3+] below which the reaction will be spontaneous in the opposite direction.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:00
1. if you have a gas at 127 degrees c, what is it's absolute temperature (kelvin)? a. 200kb. 300kc. 400kd. 500k2. if you had a gas whose absolute temperature measured 45 k, what is that temperature in celsius? a. -228 cb. -300 cc. 125 cd. 112 c
Answers: 2

Chemistry, 22.06.2019 13:00
How many moles of sulfur dioxide are produced when 4.38 moles of oxygen completely react with iron (iv) sulfide
Answers: 2

Chemistry, 23.06.2019 01:00
You wish to prepare a buffer consisting of acetic acid and sodium acetate with a total acetic acetate plus acetate concentration of 250 mm and a ph of 5. what concentrations of acetic acid and sodium acetate should you use
Answers: 1

You know the right answer?
The following reaction is spontaneous as written when the components are in their standard states:
Questions

Mathematics, 09.05.2021 20:20


Mathematics, 09.05.2021 20:20



Mathematics, 09.05.2021 20:20


History, 09.05.2021 20:20

Mathematics, 09.05.2021 20:20


Computers and Technology, 09.05.2021 20:20


English, 09.05.2021 20:20


Mathematics, 09.05.2021 20:20

Mathematics, 09.05.2021 20:20



Mathematics, 09.05.2021 20:20

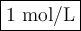
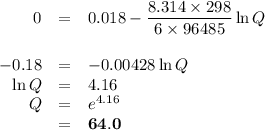
![\begin{array}{rcl}Q & = & \dfrac{\text{[Zn$^{2+}$]$^{3}$}}{\text{[Cr}^{3+}]^{2}}\\\\64.0 & = & \dfrac{4^{3}}{\text{[Cr}^{3+}]^{2}}\\\\\text{[Cr}^{3+}]^{2}& = & \dfrac{64}{64.0}\\\\& = & 1\\\text{[Cr}^{3+}] & = & \textbf{1 mol/L}\\\end{array}\\\text{[Cr$^{3+}$] must be less than $\large \boxed{\textbf{1 mol/L}}$ for the reaction to be spontaneous in the reverse}\\\text{direction.}](/tpl/images/0547/6685/5fcfe.png)


