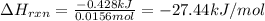Instant cold packs, often used to ice athletic injuries on the field, contain ammonium nitrate and water separated by a thin plastic divider. When the divider is broken, the ammonium nitrate dissolves according to the following endothermic reaction: NH4NO3(s)>NH+4(aq)+NO-3(aq) In order to measure the enthalpy change for this reaction, 1.25 g of NH4NO3 is dissolved in enough water to make 25.0 mL of solution. The initial temperature is 25.8 degree C and the final temperature (after the solid dissolves) is 21.9 degree C.
A. Calculate the change in enthalpy for the reaction in kilojoules per mole. (Use 1.0g/mL as the density of the solution and 4.18J/g degree C as the specific heat capacity.)
Express your answer to two significant figures and include the appropriate units.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:30
Margaret wants to make an orange flavored drink by stirring powdered drink mix into a glass of water. she doesn't like drinks that have small clumps of powdered solid in them, so she wants the drink to be a perfect solution. what factors should margaret not consider when deciding how much powder to add to her glass of water?
Answers: 3

Chemistry, 22.06.2019 12:00
Why are people not able to skip a dive to the deepest part of the ocean
Answers: 1

Chemistry, 23.06.2019 04:20
Malia was able to make a paper clip float on the surface of water what will most likely happen to the paper clip if a drop of dishwashing detergent is added near it
Answers: 1

Chemistry, 23.06.2019 07:30
How many moles of aluminum chloride are produced from 2.5 moles of magnesium chloride
Answers: 1
You know the right answer?
Instant cold packs, often used to ice athletic injuries on the field, contain ammonium nitrate and w...
Questions

Mathematics, 27.06.2020 02:01

Mathematics, 27.06.2020 02:01



Mathematics, 27.06.2020 02:01






Mathematics, 27.06.2020 02:01

Mathematics, 27.06.2020 02:01

Mathematics, 27.06.2020 02:01


Mathematics, 27.06.2020 02:01

Health, 27.06.2020 02:01

Mathematics, 27.06.2020 02:01



Mathematics, 27.06.2020 02:01

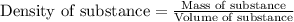
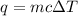
 = change in temperature =
= change in temperature = 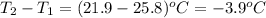
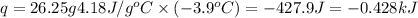
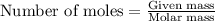
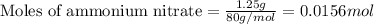
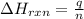
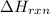 = enthalpy change of the reaction
= enthalpy change of the reaction