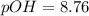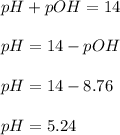Chemistry, 12.03.2020 05:54 natalie2sheffield
A chemist titrates 160.0mL of a 0.3403M aniline C6H5NH2 solution with 0.0501M HNO3 solution at 25°C . Calculate the pH at equivalence. The pKb of aniline is 4.87 . Round your answer to 2 decimal places. Note for advanced students: you may assume the total volume of the solution equals the initial volume plus the volume of HNO3 solution added.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Summarize possible ways in which phases of matter could combine to form a solution.
Answers: 2


Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2

Chemistry, 22.06.2019 16:00
How do dying stars contribute to the formation of planets
Answers: 1
You know the right answer?
A chemist titrates 160.0mL of a 0.3403M aniline C6H5NH2 solution with 0.0501M HNO3 solution at 25°C...
Questions

Arts, 06.07.2019 16:00




Arts, 06.07.2019 16:00

Spanish, 06.07.2019 16:00

Mathematics, 06.07.2019 16:00

Mathematics, 06.07.2019 16:00

Computers and Technology, 06.07.2019 16:00


Social Studies, 06.07.2019 16:00

English, 06.07.2019 16:00

Geography, 06.07.2019 16:00


Mathematics, 06.07.2019 16:00

Physics, 06.07.2019 16:00




Mathematics, 06.07.2019 16:00

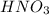
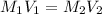
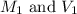 are the initial molarity and volume of
are the initial molarity and volume of 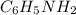 .
.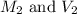 are the final molarity and volume of
are the final molarity and volume of 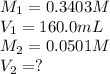
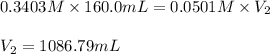
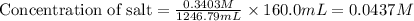
![pOH=\frac{1}{2}[pK_w+pK_b+\log C]](/tpl/images/0544/5978/6da1c.png)
![pOH=\frac{1}{2}[14+4.87+\log (0.0437)]](/tpl/images/0544/5978/4080f.png)