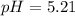Chemistry, 11.03.2020 17:05 silverdays1215
20 mL of an approximately 10% aqueous solution of ethylamine, CH3CH2NH2, is titrated with 0.3000 M aqueous HCl. Which indicator would be most suitable for this titration? The pKa of CH3CH2NH3 + is 10.75

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 17:00
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 3

Chemistry, 22.06.2019 18:00
An object displaces 652 ml of water. the volume of the object is: 0.652 cm³ 6.52 cm³ 65.2 cm³ 652 cm³
Answers: 3

Chemistry, 22.06.2019 20:30
Water undergoes a large change in density at 0 ∘ c as it freezes to form ice. calculate the percent change in density that occurs when liquid water freezes to ice at 0 ∘ c given that
Answers: 2

Chemistry, 23.06.2019 01:30
How does the attraction between particles affect the ability of a solvent to dissolve in a substance
Answers: 1
You know the right answer?
20 mL of an approximately 10% aqueous solution of ethylamine, CH3CH2NH2, is titrated with 0.3000 M a...
Questions


Mathematics, 27.09.2019 01:00

History, 27.09.2019 01:00




Arts, 27.09.2019 01:00






History, 27.09.2019 01:00







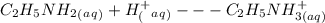
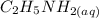 = 10%
= 10%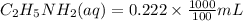
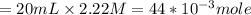
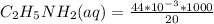
![pH= \frac{1}{2}pKa - \frac{1}{2}pKb-\frac{1}{2}log[C]](/tpl/images/0542/9187/7d506.png)
![pH = \frac{14}{2}-\frac{3.25}{2}-\frac{1}{2}log [2.22]](/tpl/images/0542/9187/6d242.png)