
Chemistry, 11.03.2020 06:06 QuestionsAnsweredNow
A mixture of CO2 and Kr weighs 41.0 g and exerts a pressure of 0.729 atm in its container. Since Kr is expensive, you wish to recover it from the mixture. After the CO2 is completely removed by absorption with NaOH(s), the pressure in the container is 0.193 atm. (a) How many grams of CO2 were originally present? (b) How many grams of Kr can you recover?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:30
98 ! and brainliest plz ! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical systems? a) water dissolves nonpolar ionic compounds. b) water dissociates ionic compounds. c) water dissociates covalent molecules. d) water dissolves nonpolar covalent substances.
Answers: 1

Chemistry, 22.06.2019 17:40
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3

Chemistry, 22.06.2019 21:30
Isopropyl alcohol, (ch3)2choh, is a common solvent. determine the percent by mass of hydrogen in isopropyl alcohol. a) 6.71% h b) 13.4% h c) 25.0% h d) 53.3% h
Answers: 1
You know the right answer?
A mixture of CO2 and Kr weighs 41.0 g and exerts a pressure of 0.729 atm in its container. Since Kr...
Questions


Biology, 26.09.2019 02:00



Mathematics, 26.09.2019 02:00

Mathematics, 26.09.2019 02:00

Spanish, 26.09.2019 02:00

Health, 26.09.2019 02:00

Biology, 26.09.2019 02:00

Health, 26.09.2019 02:00

Physics, 26.09.2019 02:00

Mathematics, 26.09.2019 02:00


Health, 26.09.2019 02:00



Mathematics, 26.09.2019 02:00



Computers and Technology, 26.09.2019 02:00

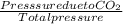
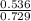
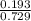
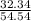 * 100 %
* 100 % 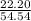 * 100 %
* 100 % 

