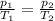Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:20
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 22.06.2019 11:00
3) in peaches, [oh]=3.16x10-11 m a) find [h+ ] b) what is the ph? c) is the solution acidic, basic, or neutral?
Answers: 1

Chemistry, 22.06.2019 14:20
Which statement explains why the bonds between non metals tend to be covalent? the bonds are found to be nondirectional they have large differences in electronegativity they have small differences in electronegativity they have ions that produce an electrostatic pull
Answers: 1

Chemistry, 22.06.2019 17:40
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1
You know the right answer?
When solving an equation using Gay-Lussac's law, which of the following steps do not apply? A. List...
Questions


Arts, 20.01.2021 22:10


Arts, 20.01.2021 22:10

Spanish, 20.01.2021 22:10

English, 20.01.2021 22:10


Computers and Technology, 20.01.2021 22:10


Mathematics, 20.01.2021 22:10

Mathematics, 20.01.2021 22:10

English, 20.01.2021 22:10



Mathematics, 20.01.2021 22:10

Biology, 20.01.2021 22:10


Mathematics, 20.01.2021 22:10

Mathematics, 20.01.2021 22:10

Mathematics, 20.01.2021 22:10