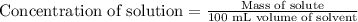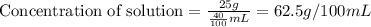Chemistry, 10.03.2020 11:28 jolleyrancher78
The concentration of a solution is equal to the mass of solute divided by the volume of solvent. Units of concentration are grams per 100 milliliters (g/100 mL, or g/dL). What is the concentration of this solution?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:00
Write a hypothesis that answers the lesson question, “while observing a chemical reaction, how can you tell which reactant is limiting? ” hypothesis: if a substance is the limiting reactant, then . . because . .
Answers: 1

Chemistry, 22.06.2019 11:00
What is the temperature of 0.750 mol of a gas stored in a 6,850 ml cylinder at 2.21 atm? . 2.95 k 5.24 k 138 k 246 k
Answers: 3


Chemistry, 22.06.2019 21:50
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1
You know the right answer?
The concentration of a solution is equal to the mass of solute divided by the volume of solvent. Uni...
Questions





Social Studies, 16.10.2020 01:01





Mathematics, 16.10.2020 01:01

Geography, 16.10.2020 01:01


Health, 16.10.2020 01:01

History, 16.10.2020 01:01

English, 16.10.2020 01:01

Mathematics, 16.10.2020 01:01

History, 16.10.2020 01:01

Mathematics, 16.10.2020 01:01


History, 16.10.2020 01:01