Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:30
If 3.00 g of titanium metal is reacted with 6.00 g of chlorine gas, cl2, to form 7.7 g of titanium (iv) chloride in a combination reaction, what is the percent yield of the product?
Answers: 1


Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 22:30
[ou.03jthe pictures below show the wavelengths and intensities of electromagnetic radiations emitted by three stars, star 1, star 2, and star 3. intensity intensity- intensity- 1000 3500 6000 8500 11000 wavelength (a) star 1 1000 3500 6000 8500 11000 1000 3500 6000 8500 11000 wavelength (a) wavelength (a) star 2 star 3 which of these statements is correct about the color of the three stars? star 2 is white in color o star 2 is yellow in color star 1 and star 3 are yellow in color star 1 and star 3 are white in color
Answers: 1
You know the right answer?
1.24 grams of magnesium phosphate tribasic dissolved in 1 L of lemon juice. What is the Ksp of the m...
Questions

Social Studies, 23.05.2020 19:00

Arts, 23.05.2020 19:00



Physics, 23.05.2020 19:00

History, 23.05.2020 19:00

Chemistry, 23.05.2020 19:00

History, 23.05.2020 19:00



Computers and Technology, 23.05.2020 19:00


Mathematics, 23.05.2020 19:00





Social Studies, 23.05.2020 19:00


Law, 23.05.2020 19:00

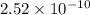
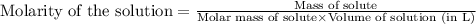
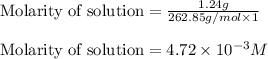
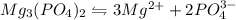
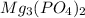 will be:
will be:![K_{sp}=[Mg^{2+}]^3[PO_4^{3-}]^2](/tpl/images/0540/2821/707f8.png)
![[Mg^{2+}]=(3\times 4.72\times 10^{-3})=1.416\times 10^{-2}M](/tpl/images/0540/2821/1537a.png)
![[PO_4^{3-}]=(2\times 4.72\times 10^{-3})=9.44\times 10^{-3}M](/tpl/images/0540/2821/66769.png)