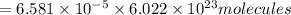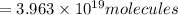Hydrogen gas, H2, reacts with nitrogen gas, N2, to form ammonia gas, NH3, according to the equation
3H2(g)+N2(g)→2NH3(g)
NOTE: Throughout this tutorial use molar masses expressed to five significant figures.
Part A
How many moles of NH3 can be produced from 18.0 mol of H2 and excess N2?
Part B
How many grams of NH3 can be produced from 4.50 mol of N2 and excess H2.
Part C
How many grams of H2 are needed to produce 12.57 g of NH3?
Part D
How many molecules (not moles) of NH3 are produced from 1.99×10−4 g of H2?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:00
When a comet collides with earth, it adds material to our planet and causes great damage. therefore, a collision like this is a a. destructive force b. constructive force c. geologic process and event d. constructive and destructive force
Answers: 1

Chemistry, 22.06.2019 04:00
Drag each label to the correct location on the chart. classify each reaction as endothermic or exothermic.
Answers: 1

Chemistry, 22.06.2019 19:30
What is the mass of oxygen gas is consumed in a reaction that produces 4.60mol so2
Answers: 3

Chemistry, 22.06.2019 20:30
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3
You know the right answer?
Hydrogen gas, H2, reacts with nitrogen gas, N2, to form ammonia gas, NH3, according to the equation<...
Questions

French, 30.11.2020 18:30


Mathematics, 30.11.2020 18:30

Arts, 30.11.2020 18:30

Social Studies, 30.11.2020 18:30

Mathematics, 30.11.2020 18:30





Mathematics, 30.11.2020 18:30

Mathematics, 30.11.2020 18:30


English, 30.11.2020 18:30

Chemistry, 30.11.2020 18:30



Computers and Technology, 30.11.2020 18:30

Health, 30.11.2020 18:30

English, 30.11.2020 18:30

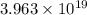 molecules of ammonia will be produced.
molecules of ammonia will be produced.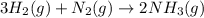
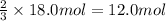 of ammonia
of ammonia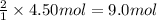 of ammonia
of ammonia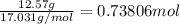
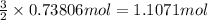 of hydrogen gas
of hydrogen gas 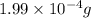
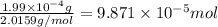
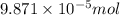 moles of hydrogen gas will give ;
moles of hydrogen gas will give ;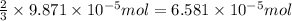 of ammonia.
of ammonia.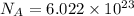 molecules/ atoms
molecules/ atoms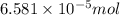 .
.