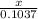Chemistry, 09.03.2020 03:47 ayowazzzgood
How many grams of magnesium oxide can be produced if 2.490 Mg react with excess oxygen?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:30
The isotonic saline solution described in part a is connected to an unknown solution via a semipermeable membrane, the unknown solution level drops. based on this information, what can be said about these two solutions?
Answers: 1

Chemistry, 21.06.2019 17:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 21.06.2019 20:00
Different isotopes indicate that an element will have different numbers of
Answers: 2

Chemistry, 22.06.2019 08:30
Which change in temperature is the smallest? a change of 1 thomson degree a change of 1 kelvin degree a change of 1 fahrenheit degree a change of 1 celsius degree
Answers: 1
You know the right answer?
How many grams of magnesium oxide can be produced if 2.490 Mg react with excess oxygen?...
Questions

Mathematics, 03.03.2021 18:50

English, 03.03.2021 18:50

Mathematics, 03.03.2021 18:50

Mathematics, 03.03.2021 18:50

English, 03.03.2021 18:50

Mathematics, 03.03.2021 18:50

Mathematics, 03.03.2021 18:50

Mathematics, 03.03.2021 18:50


Mathematics, 03.03.2021 18:50

Mathematics, 03.03.2021 18:50

History, 03.03.2021 18:50



Physics, 03.03.2021 18:50



Mathematics, 03.03.2021 18:50

Mathematics, 03.03.2021 18:50

Chemistry, 03.03.2021 18:50

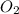 ⇒ 2MgO
⇒ 2MgO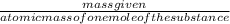
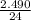
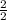 =
=