
Chemistry, 07.03.2020 06:02 perezanthony2403
Consider three generic acids: HX , HY , and HZ . The number of H plus and X minus ions in solution is equal to the initial number of HX molecules added to the solution. One quarter of the HZ molecules form H plus and Z minus ions in solution. The other three quarters of the molecules remain as HZ in solution. One half of the HY molecules form H plus and Y minus ions in solution. The other half of the molecules remain as HY in solution. Arrange the acids according to strength.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Idon't really understand this can you me and show your work.☺☺[ chemistry b] subject [ electron transfer in lonic bonds]grade( 12)
Answers: 1

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 20:00
Acm ruler with main graduations from 1 to 10 from left to right there are 10 secondary graduations between each of the main graduations there is a line that begins. at the left end of the ruler 10 secondary graduations to the left of the “1 main graduation the right end of the line ends on the eighth secondary graduation to the right of 3 how long is the line
Answers: 1

You know the right answer?
Consider three generic acids: HX , HY , and HZ . The number of H plus and X minus ions in solution i...
Questions

Advanced Placement (AP), 03.11.2020 03:40


Mathematics, 03.11.2020 03:40









Biology, 03.11.2020 03:40


History, 03.11.2020 03:40

Computers and Technology, 03.11.2020 03:40

Mathematics, 03.11.2020 03:40

Chemistry, 03.11.2020 03:40


Mathematics, 03.11.2020 03:40

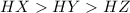
![pH=-\log[H^+]](/tpl/images/0538/0611/cf945.png)
![pH\propto \frac{1}{[H^+]}](/tpl/images/0538/0611/7625e.png)
![[H^+]_x=[HX]](/tpl/images/0538/0611/f1feb.png)
![[H^+]_y=\frac{1}{2}[HY]](/tpl/images/0538/0611/407f2.png)
![[H^+]_z=\frac{1}{4}[HZ]](/tpl/images/0538/0611/f9fc8.png)
![[H^+]_x[H^+]_y[H^+]_z](/tpl/images/0538/0611/5f37b.png)


