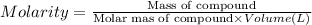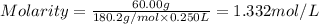Chemistry, 07.03.2020 04:59 forschoolok123456789
A student prepares a solution by dissolving 60.00 g of glucose (molar mass 180.2 g mol-1) in enough distilled water to make 250.0 mL of solution. The molarity of the solution should be reported as
a. 12.01 M
b. 12.0 M
c. 1.332 M
d. 1.33 M
e. 1.3 M

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Often on a topographic map, every fifth contour line is darkened. what is this line called? a. key b.slope c.benchmark d. index contour
Answers: 1

Chemistry, 22.06.2019 14:30
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2

Chemistry, 22.06.2019 18:30
Two people each hold the end of a rope and create waves by moving their arms up and down. this wave is best classified as a transverse wave because a) both the rope particles and the wave are moving in the same direction. b) the wave is moving up and down as the particles of the rope move horizontally. c) the wave is moving horizontally as the particles of the rope move up and down. eliminate d) the wave is moving in a parallel direction with the motion of the person's arms.
Answers: 3

Chemistry, 22.06.2019 20:10
Suppose you mix one mole of sulfuric acid (h2so4) with 1 mole of sodium hydroxide(naoh). why does the ph of the solution remain below 7? ( explain so i can get better understanding! )
Answers: 2
You know the right answer?
A student prepares a solution by dissolving 60.00 g of glucose (molar mass 180.2 g mol-1) in enough...
Questions

History, 19.09.2019 06:01




Mathematics, 19.09.2019 06:01


Social Studies, 19.09.2019 06:01


Physics, 19.09.2019 06:01


History, 19.09.2019 06:01



Biology, 19.09.2019 06:01

Business, 19.09.2019 06:01

Biology, 19.09.2019 06:01

Mathematics, 19.09.2019 06:01


English, 19.09.2019 06:01

Computers and Technology, 19.09.2019 06:01