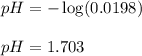Chemistry, 07.03.2020 05:25 suttonfae336
Monochloroacetic acid (HC2H2ClO2) is a skin irritant that is used in "chemical peels" intended to remove the top layer of dead skin from the face and ultimately improve the complexion. The value of Ka for monochloroacetic acid is 1.35 ✕ 10−3. Calculate the pH of a 0.31 M solution of monochloroacetic acid.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:00
Which is mostly likely why many scientists reject the cold fusion theory
Answers: 1

Chemistry, 22.06.2019 05:20
Temperature is _related to the average kinetic energy of a gas. inversely directly not disproportionally
Answers: 1

Chemistry, 22.06.2019 06:30
Summarize possible ways in which phases of matter could combine to form a solution.
Answers: 2

Chemistry, 22.06.2019 17:00
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2
You know the right answer?
Monochloroacetic acid (HC2H2ClO2) is a skin irritant that is used in "chemical peels" intended to re...
Questions

English, 29.01.2020 06:41

Chemistry, 29.01.2020 06:41


Biology, 29.01.2020 06:42




Computers and Technology, 29.01.2020 06:42





Social Studies, 29.01.2020 06:42






Mathematics, 29.01.2020 06:42

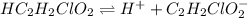
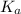 for above equation follows:
for above equation follows:![K_a=\frac{[H^+][C_2H_2ClO_2^-}}{[HC_2H_2ClO_2]}](/tpl/images/0537/9344/a55de.png)
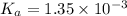
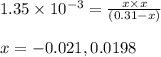
![pH=-\log[H^+]](/tpl/images/0537/9344/cf945.png)