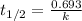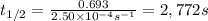Chemistry, 07.03.2020 04:45 Gghbhgy8716
The decomposition of N2O5 is described by the following equation. 2N2O5(g) → 4NO2(g) + O2(g) If the rate constant is 2.50 × 10−4 s−1, what is the half-life of this reaction?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:10
Nitrogen (n), phosphorus (p), and potassium (k) are the main nutrients in plant fertilizers. according to an industry convention, the numbers on the label refer to the mass percents of n, p2o5, and k2o, in that order. calculate the n: p: k ratio of a 30: 10: 10 fertilizer in terms of moles of each element, and express it as x: y: 1.0.
Answers: 1

Chemistry, 21.06.2019 23:50
Working with si (metric) units for each of the following commonly used measurements, indicate its symbol. liter gram milliliter kilogram meter centigram milligram centimeter kilometer second millimeter milliseconds
Answers: 1

Chemistry, 22.06.2019 10:00
In a water molecule, hydrogen and oxygen are held together by a(an) bond. a) double covalent b) ionic c) nonpolar covalent d) hydrogen e) polar covalent
Answers: 1

Chemistry, 22.06.2019 17:50
You exhale co2 which is produced during cellular respiration. co2 combines with the water in your blood's plasma to make up one half of the body's most important buffer pair, carbonic acid. the more physical activity you engage in, the more co2 your body is producing. you can see this by putting some of the cabbage indicator in a glass and then blowing bubbles into it through a straw. can you see a change in the color of the indicator?
Answers: 2
You know the right answer?
The decomposition of N2O5 is described by the following equation. 2N2O5(g) → 4NO2(g) + O2(g) If the...
Questions






Mathematics, 14.07.2020 20:01


Business, 14.07.2020 20:01



Computers and Technology, 14.07.2020 20:01



Mathematics, 14.07.2020 20:01



Mathematics, 14.07.2020 20:01

Mathematics, 14.07.2020 20:01


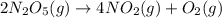
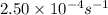
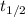 ) and rate constant (k)are related by :
) and rate constant (k)are related by :