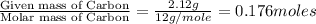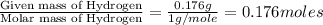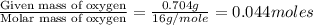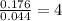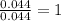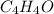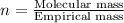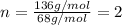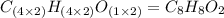Chemistry, 07.03.2020 03:46 dakotaadkins20
3.00 g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have a molecular molar mass of 136. g/mol, is burned completely in excess oxygen, and the mass of the products carefully measured product mass water .59 g Use this information to find the molecular formula of X.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 12:30
4nh3+5o2--> 4no+6h20what is the total number of moles of h2o produced when 12 mole of nh3 is completely consumed?
Answers: 3

Chemistry, 22.06.2019 13:30
Which of the following natural processes is most likely to support the formation of an underwater sinkhole? a pollution buildup from deposited minerals b limestone cave collapsing due to changes in sea level c erosion of large amounts of sand moved by ocean waves d oxidation of rock formed by chemical weathering
Answers: 1

Chemistry, 22.06.2019 19:00
Which statement best describes what happens when molecular compounds melt
Answers: 1

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1
You know the right answer?
3.00 g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have...
Questions



History, 14.05.2021 15:40


History, 14.05.2021 15:40

Biology, 14.05.2021 15:40

SAT, 14.05.2021 15:50




History, 14.05.2021 15:50

History, 14.05.2021 15:50

Social Studies, 14.05.2021 15:50

History, 14.05.2021 15:50




Mathematics, 14.05.2021 15:50


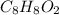
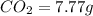
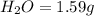
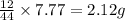 of carbon will be contained.
of carbon will be contained.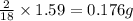 of hydrogen will be contained.
of hydrogen will be contained.